PHYSICS BYTES
Rank Booster Test Series - 04
TOPIC : Thermal Properties of Matter, Thermodynamics, Kinetic Theory of Gases
BEWARE OF NEGATIVE MARKING
1.
The two ends of a metal rod are maintained at temperature $100^{\circ}C$ and $110^{\circ}C$ The rate of heat flow in the rod is found to be $4.0~J/s$. If the ends are maintained at temperatures $200^{\circ}C$ and $210^{\circ}C$, the rate of heat flow will be:
2.
It is hotter for the same distance over the top of a fire than it is in the side of it, mainly because:
3.
In a closed room, heat transfer takes place by:
4.
According to Newton's law of cooling, the rate of cooling of a body is proportional to $(\Delta\theta)^{n},$ where $\Delta\theta$ is the difference of the temperature of the body and the surroundings, and n is equal to
5.
The maximum energy in thermal radiation from a source occurs at the wavelength 4000Å. The effective temperature of the source is:
6.
A 15 g mass of nitrogen gas is enclosed in a vessel at a temperature $27^{\circ}C$ Amount of heat transferred to the gas, so that rms velocity of molecules is doubled is about [Take $R=8.3~J/K$ mole]
7.
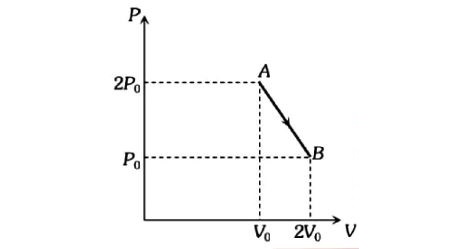
'n' moles of an ideal gas undergoes a process $A\rightarrow B$ as shown in the figure. The maximum temperature of the gas during the process will be:
8.
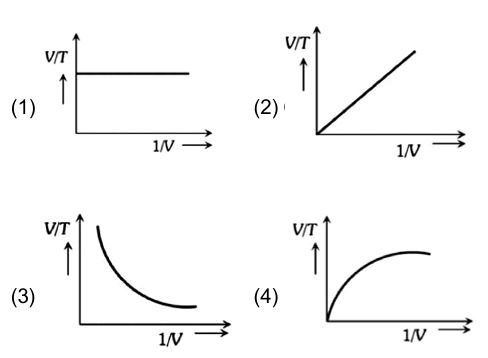
Which one of the following graphs is correct at constant pressure:
9.
The diameter of oxygen atom is 3A. The fraction of molecular volume of the actual volume occupied by oxygen at STP is:
10.
One mole of an ideal gas at an initial temperature of T K does 6 R joules of work adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is $5/3$ the final temperature of gas will be:
11.
Air in a cylinder is suddenly compressed by a piston, which is then maintained at the same position. With the passage of time:
12.
The specific heat of a gas in an isothermal process is:
13.
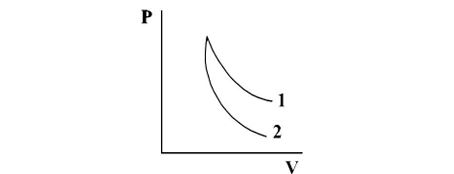
P-V plots for two gases during adiabatic process are shown in figure. Plots 1 and plots 2 should correspond respectively to:
14.
Steam at $100^{\circ}C$ is passed into 20 g of water at $10^{\circ}C.$ When water acquires a temperature of $80^{\circ}C$ the mass of water present will be [Take specific heat of water = 1 cal $gm^{-1}C^{-1}$ and latent heat of steam = 540 cal g⁻¹]
15.
A one litre flask contains certain quantity of mercury. If the volume of air inside the flask remains the same at all temperatures then the volume of mercury in the flask is (volume expansion coefficient of mercury is 20 times that of flask):
16.
The coefficient of volume expansion of glycerin is $49\times10^{-5}K^{-1}$ What is the fractional change in its density for a $30^{\circ}C$ rise in temperature :
17.
We plot the graph having temperature °C on x-axis and in °F on y-axis. If the graph is straight line, then the correct statement is :
18.
Three rods of same dimensions have thermal conductivities 3K, 2K and K. They are arranged as shown, with their ends at $100^{\circ}C$ $50^{\circ}C$ and $0^{\circ}C$. The temperature of their junction is
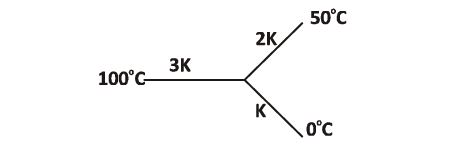
19.
In a vertical U-tube containing a liquid, the two arms are maintained at different temperatures, $t_{1}$ and $t_{2}$. The liquid columns in the two arms have heights $l_{1}$ and $l_{2}$ respectively. The coefficient of volume expansion of the liquid is equal to:
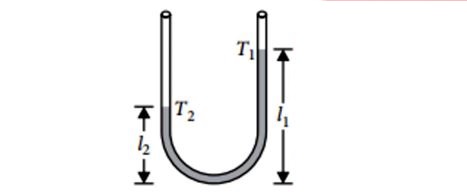
20.
Two holes of unequal diameters $d_{1}$ and $d_{2}(d_{1}>d_{2})$ are cut in a metal sheet. If the sheet is heated:
21.
A solid with coefficient of linear expansion $\alpha$ just floats in a liquid whose coefficient of volume expansion is $\gamma$. If the system is heated the solid will
22.
The volume of the bulb of a mercury thermometer at $0^{\circ}C$ is $V_{0}$ and cross-section of the capillary is $A_{0}.$ The coefficient of linear expansion of glass is $\alpha_{g}$ per °C and the cubical expansion of mercury $\gamma_{m}$ per °C. If the mercury just fills the bulb at $t=0^{\circ}C$, what is the length of mercury column in capillary at $t^{\circ}C$:
23.
What is the final temperature when 200 g of ice at $-20^{\circ}C$ is dropped into 350 g of water at $40^{\circ}C$ contained in a calorimeter of 50g water equivalent:
24.
If R = universal gas constant, the amount of heat needed to raise the temperature of 2 mole of an ideal monoatomic gas from 273K to 373K when no work is done
25.
A vessel containing 5 litres of a gas at 0.8 m pressure is connected to an evacuated vessel of volume 3 litres. The resultant pressure inside will be (assuming whole system to be isolated)
26.
In an isothermal process the volume of an ideal gas is halved. One can say that
27.
A gas for which $\gamma=1.5$ is suddenly compressed to $\frac{1}{4}$ th of the initial volume. Then the ratio of the final to the initial pressure is
28.
In an adiabatic change, the pressure P and temperature T of a monoatomic gas are related by the relation $P\propto T^{c}$ where c equals
29.
Three samples of the same gas A, B and $C(\gamma=3/2)$ have initially equal volume. Now the volume of each sample is doubled. The process is adiabatic for A isobaric for B and isothermal for C. If the final pressures are equal for all three samples, the ratio of their initial pressures are
30.
In pressure-volume diagram given below, the isochoric, isothermal, and isobaric parts respectively, are
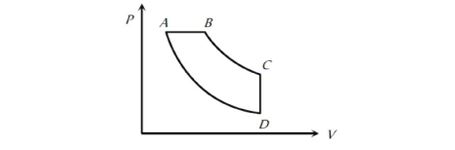
31.
An electric heater supplies heat to a system at a rate of 100 W. If system work at a rate of 75 jules per second. At what rate is the internal energy increase
32.
A geyser heats water flowing at the rate of 3.0 liters per minute from $27^{\circ}C$ to $77^{\circ}C$ If the geyser operates on a gas burner, what is the rate of consumption of the fuel if its heat of combustion is $4.0\times10^{4}J/g$?
33.
The mean free path is average distance covered by a molecule between two successive collision.
34.
All of the following are properties of ideal gases except:
35.
How many diagrams showing relationships between pressure, volume, and temperature below is/are incorrect for ideal gasses ?
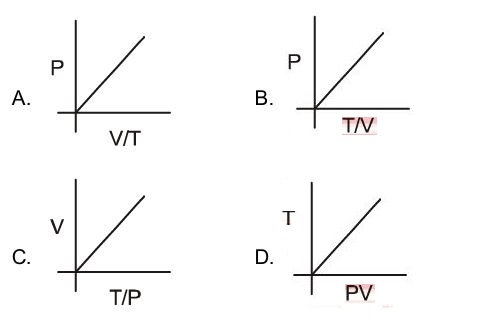
36.
An inflated rubber balloon contains one mole of an ideal gas, has a pressure P, volume V and temperature T. If the temperature rises to 1.1 T, and the volume is increased to 1.05 V, the final pressure will be:
37.
Calculate the ratio of the mean free paths of the molecules of two gases having molecular diameters 1Å and 2 Å. The gases may be considered under identical conditions of temperature, pressure and volume.
38.
The energy of a gas/litre is 300 joules, then its pressure will be :
39.
Value of $\gamma$ is :
40.
1 mole of a gas with $\gamma=7/5$ is mixed with 1 mole of a gas with $\gamma=5/3$, then the value of $\gamma$ for the resulting mixture is
41.
Assertion: The total translational kinetic energy of all the molecules of a given mass of an ideal gas is 1.5 times the product of its pressure and its volume.
Reason: The molecules of a gas collide with each other and the velocities of the molecules change due to the collision.
Reason: The molecules of a gas collide with each other and the velocities of the molecules change due to the collision.
42.
An ideal monoatomic gas is taken through the thermodynamic states $A\rightarrow B\rightarrow C\rightarrow D$ via the paths shown in the figure. If $U_{A}$, $U_{B}$, $U_{c}$ and $U_{D}$ represent the internal energy of the gas in state A, B, C and D respectively, then which of the following is not true?
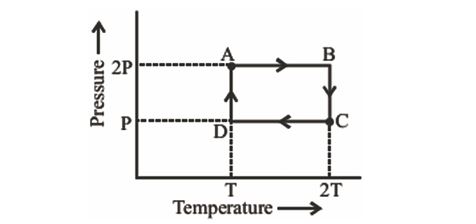
43.
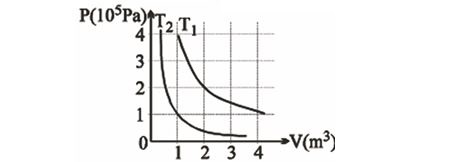
The following graphs shows two isotherms for a fixed mass of an ideal gas. The ratio of r.m.s. speed of the molecules at temperatures $T_{1}$ and $T_{2}$ is
44.
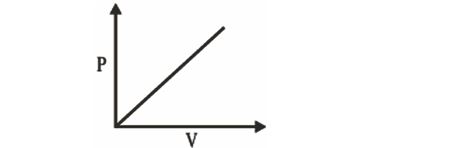
1 mole of monoatomic ideal gas is expanded as per following process. What will be molar heat capacity?
45.
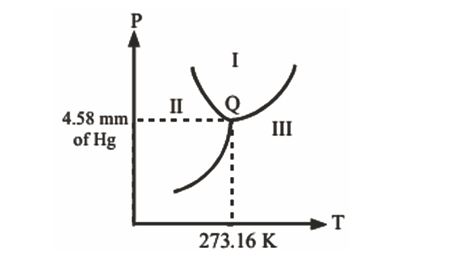
In the phase diagram shown, the point Q corresponds to the triple point of water. The regions I, II and III respectively correspond to phase