PHYSICS BYTES
Rank Booster Test Series - 12
TOPIC : Dual Nature of Radiation and Matter, Atoms, Nuclei, Semiconductor Electronics
BEWARE OF NEGATIVE MARKING
1.
A photon collides inelastically with a stationary hydrogen atom in the ground state. Energy of the colliding photon is 10.2 eV. After a time interval of the order of microsecond, another photon collides with the same hydrogen atom inelastically with an energy of 15 eV. What will be observed by the detector?
2.
For a hydrogen-like atom, the angular momentum of the electron in the nth orbit is given by $L_{n}=n^{2}\hbar.$ The dependence of the radius of the $n^{th}$ orbit on n is:
3.
Consider the following statements:
$S_{1}$ In the photoelectric effect, the maximum kinetic energy of emitted electrons depends only on the frequency of incident radiation, not on its intensity.
$S_{2}$ In the Bohr model of the hydrogen atom, the energy difference between successive orbits decreases as the principal quantum number increases.
$S_{3}$ In nuclear reactions, the binding energy per nucleon generally increases with mass number up to iron.
$S_{4}$ In a semiconductor, increasing temperature increases electrical conductivity due to an increase in charge carriers.
Which of the above statements are correct?
$S_{1}$ In the photoelectric effect, the maximum kinetic energy of emitted electrons depends only on the frequency of incident radiation, not on its intensity.
$S_{2}$ In the Bohr model of the hydrogen atom, the energy difference between successive orbits decreases as the principal quantum number increases.
$S_{3}$ In nuclear reactions, the binding energy per nucleon generally increases with mass number up to iron.
$S_{4}$ In a semiconductor, increasing temperature increases electrical conductivity due to an increase in charge carriers.
Which of the above statements are correct?
4.
In discharge tube experiment electrons are created by Thermionic emission and electrons are moving by applying potential difference of 49V. Find de-Broglie wavelength associated by electron:
5.
The two lines A and B shown in Figure are the graphs of the de Broglie wavelength as a function of $1/\sqrt{V}$ (V is the accelerating potential) for two particles having the same charge. Which of the represents the particle of heavier mass:
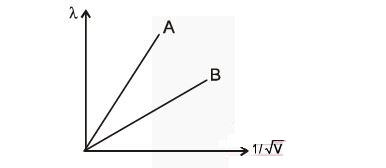
6.
Two metals A and B have work functions 4eV and 10 eV respectively. Which metal has higher threshold wave length:
7.
A photon of energy 4 eV is incident on a metal surface whose work function is 2 eV. The minimum reverse potential to be applied for stopping the emission of electrons is:
8.
Find de Broglie wavelength of a neutron at $127^{\circ}C.$ Given Boltzmann constant, $k_{B}=1.38\times10^{-23}J/K$ :
9.
If the momentum of electron is changed by P, then the de-Broglie wavelength associated with it changes by 0.4%. The initial momentum of electron will be :
10.
Two photons having
11.
A point source of light is placed at a distance of 1.0 m from a caesium photocell and under saturation conditions the number of emitted photo electrons is n. If the source is now placed 0.5 m from the cell, the number of emitted electrons will be:
12.
Work function of aluminium metal is 4.2 electron volt. If two photons having energies 4 electron volt and 3 electron volts falls on the aluminium metal then which option is correct:
13.
Photocells are used for the
14.
Let $v_{1}$ be the frequency of the series limit of the Lyman series, $v_{2}$ be the frequency of the first line of the Lyman series, and $v_{3}$ be the frequency of the series limit of the Balmer series:
15.
The ionisation potential of H-atom is 13.6 V. When it is excited from ground state by monochromatic radiations of 970.6 Å, the number of emission lines will be (according to Bohr's theory):
16.
The wavelength of radiation is $\lambda_{0}$ when an electron jumps from third to second orbit of hydrogen atom. For the electron to jump from the fourth to the second orbit of the hydrogen atom, the wavelength of radiation emitted will be :
17.
An electron is revolving around a nucleus of hydrogen atom in the first orbit. The radius of this orbit is 0.53 Å:
18.
A particular hydrogen like atom has its ground state binding energy 122.4 eV. Then which is incorrect:
19.
An electron in a hydrogen like atom is in excited state and has total energy equal to -3.4 eV. The de-Broglie wavelength of electron is :
20.
Imagine an atom made up of a proton and a hypothetical particle of double the mass of the electron but having the same charge as the electron. Apply the Bohr atom model and consider all possible transitions of this hypothetical particle to the first excited level. The longest wavelength of photon that will be emitted has wavelength $\lambda$ (given in terms of Rydberg constant R for the hydrogen atom) equal to:
21.
An $\alpha$-particle having energy 10 MeV travelling directly towards the nucleus of an atom is deflected back when its distance from the nucleus is $4\times10^{-14}$ m. Calculate the atomic number of the atom:
22.
In a hypothetical fission reaction: ${}_{92}^{236}X\rightarrow {}^{141}Y + {}_{36}Z + 3_{0}^{1}n$
Calculate the total energy released per nuclear fission in MeV units, Given mass of neutron = 1.009 a.m.u. mass of X-nucleus = 235.891 a.m.u., mass of Y-nucleus = 140.673 a.m.u., mass of Z-nucleus = 91.791 a.m.u.
Calculate the total energy released per nuclear fission in MeV units, Given mass of neutron = 1.009 a.m.u. mass of X-nucleus = 235.891 a.m.u., mass of Y-nucleus = 140.673 a.m.u., mass of Z-nucleus = 91.791 a.m.u.
23.
Two nucleons are at a separation of 1 fermi. The net force between them is $F_{1}$ if both are neutrons, $F_{2}$ if both are protons, and $F_{3}$ if one is a proton and the other is a neutron:
24.
The ionisation energy of hydrogen atom is 13.6 eV. Following Bohr's theory of atom, the energy required for removing an electron from the first orbit to the second orbit of He ion is :
25.
The speed of an electron in the first exited state of hydrogen atom is:
26.
The wavelength of series limit of Lyman series is:
27.
During a nuclear fission reaction,
28.
For nuclei with $A>150$ :
(a) The binding energy of the nucleus increases on an average as A increases
(b) The binding energy per nucleon decreases on an average as A increases
(c) If the nucleus breaks into two roughly equal parts, energy is released
(d) If two nuclei fuse to form a bigger nucleus, energy is released
(a) The binding energy of the nucleus increases on an average as A increases
(b) The binding energy per nucleon decreases on an average as A increases
(c) If the nucleus breaks into two roughly equal parts, energy is released
(d) If two nuclei fuse to form a bigger nucleus, energy is released
29.
The correct curve showing variation of binding energy per nucleon(B) with mass number (A) of the nucleus is:
30.
The curve of binding energy per nucleon as a function of atomic mass number has a sharp peak for helium nucleus. The implies that helium
31.
The wavelength of the first line of Balmer series of hydrogen atom is $\lambda$. The wavelength of this line of doubly ionized lithium atom $(z=3)$ is:
32.
The graph given below represents the I-V characteristics of a Zener diode. Which part of the characteristics curve is most relevant for its operation as a voltage regulator:
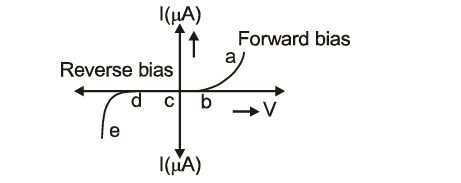
33.
A p-n junction diode is connected to a battery of emf 5.5V and external resistance 5.1 $k\Omega$. The barrier potential in the diode is 0.4V. The current in the circuit is:
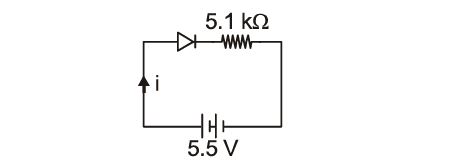
34.
A Zener diode has a contact potential of 1V in the absence of biasing. It undergoes Zener breakdown for an electric field of $10^{6}V/m$ at the depletion region of p-n junction. If the width of the depletion region is $2.5\mu m$ what should be the reverse biased potential for the Zener breakdown to occur:
35.
The reverse saturation current of p-n diode:
36.
The peak voltage in the output of a half-wave diode rectifier fed with a sinusoidal singal without filter is 10V. The DC component of the output voltage is:
37.
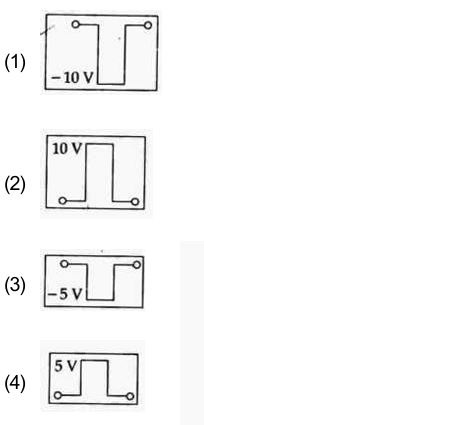
If in a p-n junction, a square input signal to 20V is applied, as shown:
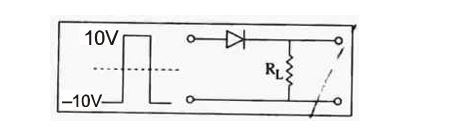 then the output across $R_{L}$ will be :
then the output across $R_{L}$ will be :
38.
At absolute zero temperature a semiconductor behaves like :
39.
When a P-N junction is unbiased, the junction current at equilibrium is:
40.
The drift current in a P-N junction is:
41.
Region which have no free electron and holes in a P-N junction is:
42.
Out of the following, in which configuration is the diode valve in forward biased:
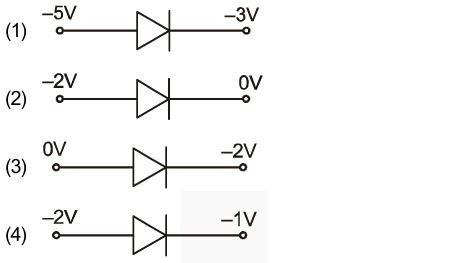
43.
Reverse bias applied to a junction diode:
44.
A 12.5 eV electron beam is used to bombared gaseous hydrogen at room temperature. It will emit:
45.
Photoelectrons are ejected from a metal when light of frequency v falls on it. Pick out the wrong statement from the following: